Molecular Descriptors For Cheminformatics Pdf To Excel
Classifications: Chemical descriptors are used to calculate and to develop methods for chemical property calculations (QSPR - quantitative structure-property relationship) or chemical activity (QSAR - quantitative structure-activity relationship) calculations. A common classification method for descriptors can be taken textbooks and a collection of common molecular descriptors in the Handbook of molecular descriptors []. 2 Chainz Based On A Tru Story Deluxe Version Download Zip. • 0D - bond counts, mol weight, atom counts • 1D - fragment counts, H-Bond acc/don, Crippen, PSA, SMARTS • 2D - topological descriptors (Balaban, Randic, Wiener, BCUT, kappa, chi) • 3D - geometrical descriptors (3D WHIM, 3D autocorrelation, 3D-Morse) + surface properties + COMFA • 4D - 3D coordinates + conformations (JCHEM conformer, CORINA, gold set, Crystaleye) Tools for descriptor calculations: A selection of commercial and free descriptor calculation utilities is collected under the molecular descriptor or the list or new programs are posted to CCL.
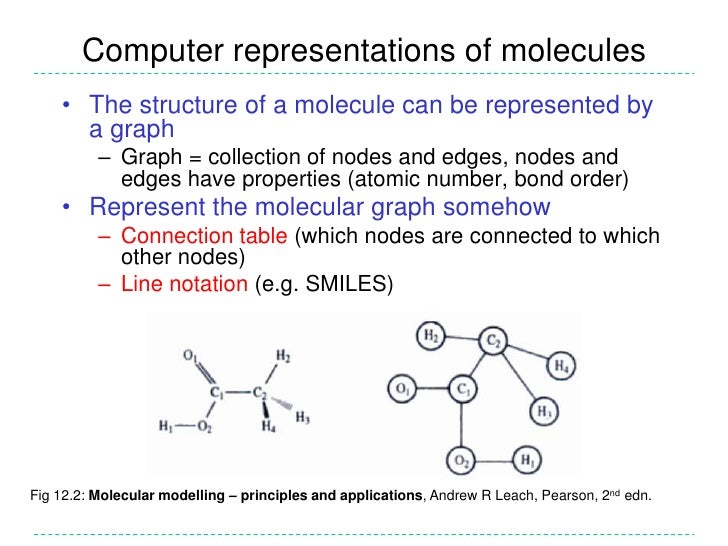
Molecular descriptors play a fundamental role in chemistry, pharmaceutical sciences, environmental protection policy, and health researches, as well as in quality control, being the way molecules, thought of as real bodies, are transformed into numbers, allowing some mathematical treatment of the chemical information contained in the molecule. This was defined by Todeschini and Consonni as: 10 Feet Super Stomper Download Itunes. ' The molecular descriptor is the final result of a logic and mathematical procedure which transforms chemical information encoded within a symbolic representation of a molecule into a useful number or the result of some standardized experiment.'
By this definition, the molecular descriptors are divided into two main categories: experimental measurements, such as,,,, and, in general, physico-chemical properties, and theoretical molecular descriptors, which are derived from a symbolic representation of the molecule and can be further classified according to the different types of molecular representation. The main classes of theoretical molecular descriptors are: 1) 0D-descriptors (i.e.
ChemAxon’s Calculator Plugin suite is a cheminformatics toolkit to calculate. Calculating molecular descriptors. Cheminformatics and Computational Chemistry in. Excel sheet, databases. All the molecular descriptors. LICSS is an Excel-based chemical spreadsheet. JChemPaint and OPSIN open source libraries to provide cheminformatics. Molecular Descriptors For Cheminformatics Pdf To Jpg. 5/28/2017 0 Comments February 13, 2016 Chemical Computing Group Announces The Winners of.
Constitutional descriptors, count descriptors), 1D-descriptors (i.e. List of structural fragments, fingerprints), 2D-descriptors (i.e. Graph invariants), 3D-descriptors (such as, for example, 3D-MoRSE descriptors, WHIM descriptors, GETAWAY descriptors, quantum-chemical descriptors, size, steric, surface and volume descriptors), 4D-descriptors (such as those derived from GRID or CoMFA methods, Volsurf). Contents • • • • • • • Invariance properties of molecular descriptors [ ] The of molecular descriptors can be defined as the ability of the algorithm for their calculation to give a descriptor value that is independent of the particular characteristics of the molecular representation, such as atom numbering or labeling, spatial reference frame, molecular conformations, etc. Invariance to molecular numbering or labeling is assumed as a minimal basic requirement for any descriptor.
Two other important invariance properties, and, are the invariance of a descriptor value to any translation or rotation of the molecules in the chosen reference frame. These last invariance properties are required for the 3D-descriptors.
Degeneracy of molecular descriptors [ ] This property refers to the ability of a descriptor to avoid equal values for different molecules. In this sense, descriptors can show no degeneracy at all, low, intermediate, or high degeneracy. For example, the number of molecule atoms and the molecular weights are high degeneracy descriptors, while, usually, 3D-descriptors show low or no degeneracy at all. • Roberto Todeschini and Viviana Consonni, Handbook of Molecular Descriptors, Wiley-VCH, 2000. Bibliography [ ] Roberto Todeschini and Viviana Consonni, Molecular Descriptors for Chemoinformatics (2 volumes), Wiley-VCH, 2009.
Mati Karelson, Molecular Descriptors in QSAR/QSPR, John Wiley & Sons, 2000. James Devillers and Alexandru T. Balaban (Eds.), Topological indices and related descriptors in QSAR and QSPR. Taylor & Francis, 2000. Lemont Kier and Lowell Hall, Molecular structure description.
Academic Press, 1999. Download Naruto Shippuden Episode 341 Indonesia Subtitle. Balaban (Ed.), From chemical topology to three-dimensional geometry. Plenum Press, 1997. External links [ ] •.